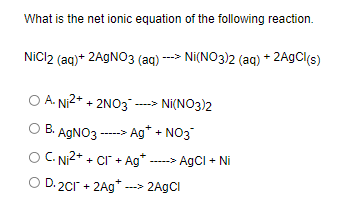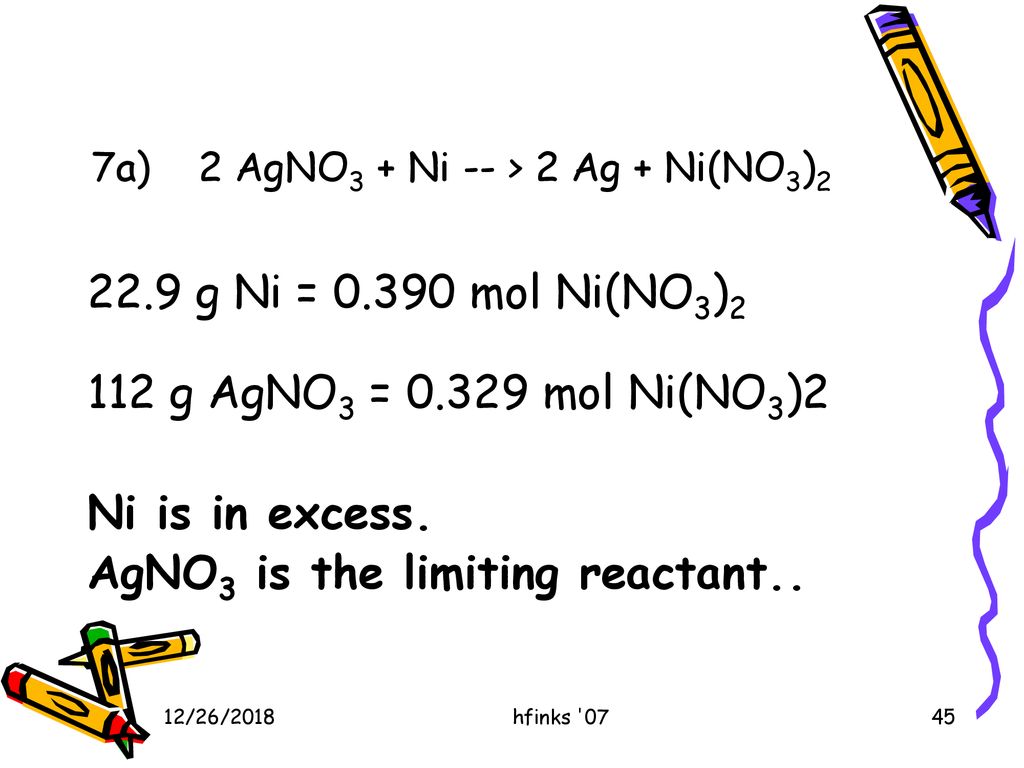Direct Growth of Ag/Ni(OH)2 Composite on Cu Foam by a Modified Galvanic Displacement Reaction Followed by Electrodeposition

SOLVED: Select the correct balanced net ionic equation for each of the following reactions occurring in aqueous solution: Cu(NO3)2 + 2NaCl -> 2AgCl + Cu(NO3)2 Ni(NO3)2 + 2AgNO3 -> 2Ag + Ni(NO3)2

Oxidation of Aniline with Silver Nitrate Accelerated by p-Phenylenediamine: A New Route to Conducting Composites | Macromolecules

_____ Zn (s) + _____ AgNO3 (aq) → _____ Zn(NO3)2 (aq) + _____ Ag (s) b. _____ N2 (g) + _____ H2 (g) → _____ - Brainly.in
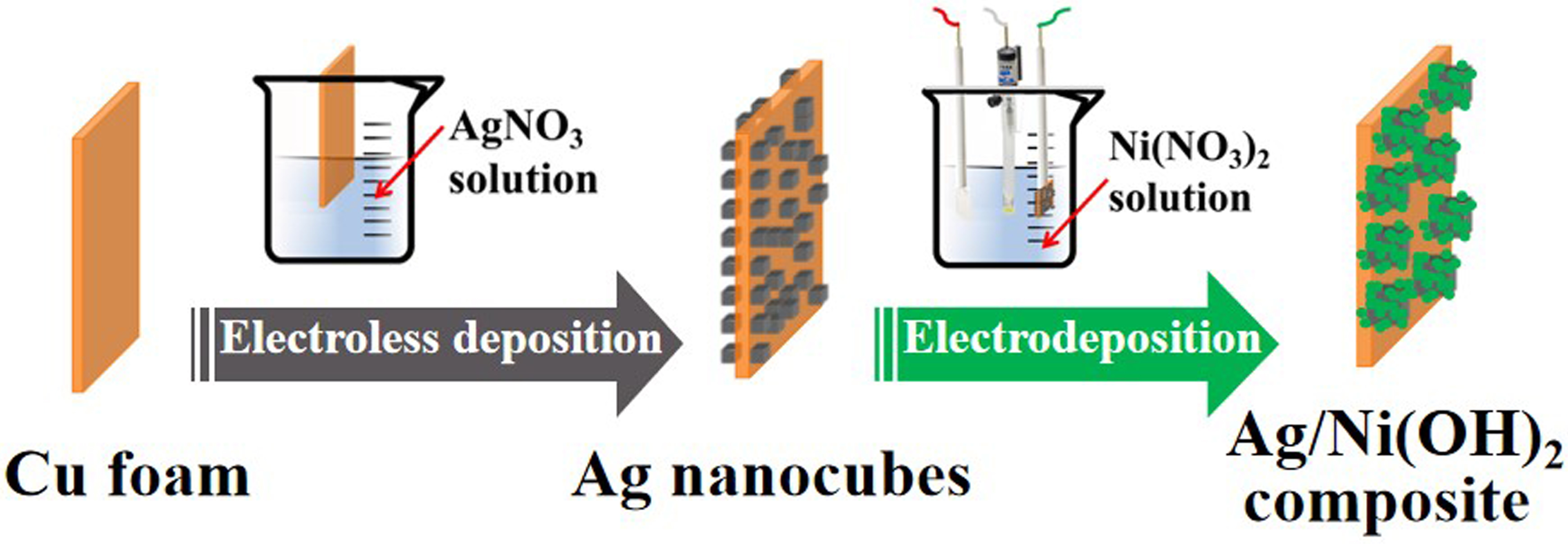
Metals | Free Full-Text | In Situ Construction of Ag/Ni(OH)2 Composite Electrode by Combining Electroless Deposition Technology with Electrodeposition

SOLVED: Nickel is a more active metal than silver; therefore, nickel will replace silver ions in a silver nitrate solution to form Nickel (II) nitrate and silver metal. How many grams of



![physical science 20] stoichiometry?????? : r/HomeworkHelp physical science 20] stoichiometry?????? : r/HomeworkHelp](https://preview.redd.it/physical-science-20-stoichiometry-v0-8nbxhcuimhqb1.jpg?auto=webp&s=a5e973323a2ae9883f568683e3d6a9c6f088002d)